ISASS Guideline – Intraosseous Ablation of the Basivertebral Nerve for…
- Home
- Public Policy
- Policy Statements
- ISASS Guideline – Intraosseous Ablation of the Basivertebral Nerve for the Relief of Chronic Low Back Pain
- Categories
- Tags
ISASS Guideline – Intraosseous Ablation of the Basivertebral Nerve for the Relief of Chronic Low Back Pain
Morgan Lorio1; Olivier Clerk-Lamalice2; Douglas P. Beall3; Terrence Julien4
1Advanced Orthopedics, Altamonte Springs, Florida; 2Beam Interventional & Diagnostic Imaging, Calgary, Canada; 3Clinical Radiology of Oklahoma, Edmond, Oklahoma; 4BayCare Medical Group, Tampa, FloridaRationale
This ISASS guideline is generated to respond to growing requests for background, supporting literature and evidence, and proper coding for intraosseous ablation of the Basivertebral nerve (BVN) for chronic low back pain (CLBP).
Introduction
Prevalence and Clinical Presentation
Low back pain is the most expensive occupational disorder in the United States and the leading cause of disability worldwide1-3. Thirty percent of Americans have low back pain (LBP) at any given time, leading to approximately 50 million physician visits in the U.S. annually. Although many of these patients improve with little to no treatment, an estimated 30 million adults in the U.S. currently suffer from chronic low back pain (CLBP), defined as pain lasting for greater than 12 weeks4-10. These CLBP patients have direct yearly costs of over $90 billion/year11. As is the case with many medical conditions, a minority of CLBP patients consume the majority of healthcare resources. Analyses of commercial payer and Medicare claims databases reveals that 15% of CLBP patients account for 75% of healthcare costs, with average claims of $24,700 over a three-year period in the high healthcare utilizer group (MarketScan, Truven Health Analytics from October 2011 to September 2016).
Disc degeneration (DD) is a strong risk factor for CLBP12-1, and the disc has been the target of many treatments. Recent scientific research has reexamined CLBP sources, and there is evidence suggesting that the disc and adjacent endplates act as one functional unit and that the vertebral endplate is a source of pathologic innervation that occurs with disc degeneration.
Indeed, the endplates must balance conflicting requirements of being strong to prevent vertebral fracture and porous to facilitate transport between disc cells and vertebral capillaries. Consequently, endplates are particularly susceptible to damage leading to inflammation and nerve proliferation.
The sensory nerves within the center of the vertebral body converge to form the basivertebral nerve (BVN)15-16. The BVN exits the vertebral body posteriorly via the basivertebral foramen before communicating with the sinuvertebral nerve then the ventral rami of the spinal nerves or by nerves derived from the gray rami communicantes16. When the density of pain fibers between normal endplates and degenerated endplates is compared, the BVN density is considerably higher in patients with degenerated endplates, further suggesting the role of pain transmission via the BVN in patients with CLBP16. The pain transmission of the endplates toward the BVN has been named of ‘vertebrogenic’ origin14, 15. Patients with vertebrogenic pain are thought to present with low back pain, with or without referral into the buttocks or thighs (somatic referred pain).
Traditional treatments for CLBP
CLBP may lead to a compromised quality of life, strained societal and familial relationships, and increased absenteeism or work-related disability claims. A lack of current validated diagnostic reference standards leads 85% of individuals to be diagnosed with non-specific low back pain. This non-specific diagnosis leads to non-specific care that follows care pathways that are not scientifically validated (Table 1). Individuals are advised to stay active, engage in core strengthening, lose weight and avoid bed rest. They are put through non-sequential, palliative injection treatments in the hope that these treatments will help the patient’s function and that the pain will then regress. For refractory cases, surgical intervention may be recommended.
Table 1. Non-surgical management may include:
1. Avoidance of activities that aggravate pain 2. Trial of Chiropractic manipulation 3. Trial of physical therapy 4. Cognitive support and recovery reassurance 5. Spine biomechanics education 6. Specific lumbar exercise program 7. Home use of heat/cold modalities 8. Low impact aerobic exercise as tolerated 9. Pharmacotherapy (e.g., non-narcotic analgesics, NSAIDs etc.) Radiologic Imaging
The MRI correlation between vertebral endplate pathology and CLBP was made by Modic et al. in 1988 who described intraosseous MRI changes adjacent to damaged vertebral endplates in individuals with CLBP.17
This correlation is based on the T1 and T2-weighted signal of the endplates. Three types of signal change have been described: Modic Change 1 (MC1), Modic Change 2 (MC2) and Modic Change 3 (MC3). MC1 corresponds to bone marrow edema and inflammation (hypointense T1-weighted signal and hyperintense T2-weighted signal) (Figures 1A and 1B). MC2 is characterized by hyperintense T1-weighted signal and hyperintense T2-weighted signal (Figures 1C and 1D) is the conversion of normal red hemopoietic bone marrow into yellow fatty marrow. MC3 is described as bone sclerosis and is characterized by hypointense T1-weighted signal and hypointense T2-weighted signal. MC1 is considered unstable and some studies have suggested this to be painful 18, 19. MC2 has been suggested to be less correlated to pain.20 Patients with MC3 change are rarely symptomatic. Some speculate that the MC1 change is caused by an inflammatory response due to fissuring and disruption between the disc and the bone that develops along with endplate microfractures18-22 while others think that some of these changes could be due to a chronic infection23. The afferent pain pathway travels from the disc and endplate to converge as the BVN before being transmitted through the dorsal root ganglion (DRG) to the central nervous system and perceived as low back pain. The initial neural convergence as the BVN in the mid-portion of the vertebral body provides a potential target for treatment. Having an MRI done prior to the patient’s consultation with chronic low back pain is essential to adequately determine the pain generator and viable treatment alternatives. Painful Modic changes most frequently affect the L4-L5 and L5-S1 levels; in fact, Kuisma et al. found a 2.28 odds ratio for the presence of Modic changes at L5-S1 in individuals with CLBP24.
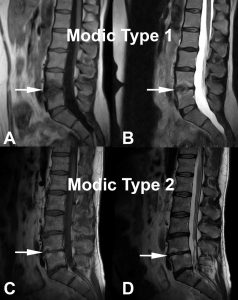
Figure 1 – MC1 and MC2 changes illustrated. A) and B) demonstrate decreased signal intensity on T1-wighted images, and increased signal intensity on T2-weighted images respectively (white arrows) corresponding to MC1. C) and D) correspond to MC2 with increased signal intensity on T1-weighted images and on T2-weighted images respectively (white arrows).
Procedure
A unilateral transpedicular approach is utilized to advance a straight introducer under fluoroscopic guidance to the juncture of the pedicle and the vertebral body. A curved cannula assembly is utilized to penetrate the vertebral body and navigate towards the basivertebral nerve, which is located in the posterior half of the vertebral body. A straight channeling stylet is then utilized to extend the channel to the midline location of the basivertebral nerve. A bipolar probe is inserted into the posterior half of the vertebral body, connected to the radiofrequency generator, and energy is applied for 15 minutes to destroy the basivertebral nerve. Once ablated, these nerves no longer transmit pain signals.
Data from the two Level I randomized controlled trials would suggest that in approximately 80% of patients, two vertebral bodies are treated, which constitute one vertebral motion segment. In the remaining patients, 1 or 2 additional vertebral bodies are treated for a total of 2-3 vertebral motion segments.
Animal studies performed as a part of a Food and Drug Administration (FDA) submission also showed that the intraosseous BVN does not regenerate, and that the vertebrae return to pre-treatment strength after a period of normal healing [personal communication of unpublished bovine research by Professor Jeffrey C. Lotz, PhD (David S. Bradford MD Endowed Chair in Orthopaedic Surgery at UCSF) and corroborated by published ovine research by Hoopes, PJ, et.al.25].
Published Literature
Becker, et al Pilot Study 26
Single-arm, open-label, first-in-human pilot study to determine the early efficacy and safety of intraosseous BVN ablation for the treatment of chronic lumbar back pain (CLBP). Seventeen patients with 6 or more months of CLBP and MC1 or MC2 changes were enrolled. Sixteen patients were successfully treated using radiofrequency energy to ablate the BVN within the vertebral bodies adjacent to the diagnosed level (based on positive discography).
The mean age of enrolled patients was 48 years. Baseline measurements of mean ODI and VAS were 52±13 (severe disability on the 0 to 100-point disability impact scale) and 61 (on the 0 to 100-point low back pain scale). Statistically significant improvements were noted in all outcome measures at 3-months. ODI decreased an average of 29 points to a mean of 23±21 at 3 months of follow-up (p<.001). This statistically significant improvement in ODI was maintained through the 12-month follow-up.
Truumees, et al Case Series27:
This study was a prospective, single-arm, multi-center, open-label study to evaluate the effectiveness of intraosseous RF ablation of the BVN for the treatment of presumed vertebrogenic related CLBP in typical spine practice settings with more permissive inclusion of typical CLBP patients (such as patients who have had prior discectomy and utilizers of extended-release narcotics). Consecutive patients with CLBP of at least 6 months duration and with MC1 or MC2 vertebral endplate changes between L3 to S1, were treated with RF ablation of the BVN in up to 4 vertebral bodies. The primary endpoint was patient reported change in ODI from baseline to 3 months post procedure. Secondary outcome measures included change in LBP pain VAS, SF-36, EQ-5D-5L, and responder rates.
The median age of patients was 45 years within the 28 patients enrolled. Baseline ODI was 48.5 and VAS was 6.36 cm (on a 0 to 10 cm scale). Seventy-five percent (75%) of the study patients reported LBP symptoms for ≥ 5 years with 25% actively using opioids and 61% previously treated with injections. Clinically meaningful and statistically significant improvements where demonstrated in all outcome measures at the 3-month primary endpoint. Mean reduction in ODI from baseline at three months post treatment was – 30.07 +14.52 points (p<0.0001). The mean reduction in VAS pain score from baseline was -3.50+2.33 (p<0.0001). Using a minimal clinically important difference (MCID) of ≥ 10-point improvement in ODI, 93% of patients were responders; using MCID of a ≥ 20-point improvement in ODI, 75% were responders. Likewise, VAS MCID of a ≥ 2.0 cm reduction was achieved in 75% of patients. Importantly, in this population of working age individuals, 83% reported improvement in work function. This nonrandomized consecutive series study demonstrated that minimally invasive RF ablation of the BVN resulted in a significant improvement in pain and function at 3 months in this population of real-world patients with chronic suspected vertebrogenic related LBP.
INTRACEPT study28:
This prospective, parallel, open-label, randomized control trial conducted at 20 US sites compared the effectiveness of intraosseous RF ablation of the BVN to standard care for the treatment of chronic low back pain (CLBP) in patients suspected to have vertebrogenic-related pain symptomatology. A total of 140 patients with CLBP of at least 6 months duration, with MC1 or MC2 vertebral endplate changes between L3 to S1, were randomized 1:1 to undergo either RF ablation of the BVN or continue standard care. The primary endpoint was a between-arm comparison of the mean change in ODI from baseline to 3 months post-treatment. Secondary outcome measures included LBP pain scores via VAS, ODI, VAS responder rates, SF-36, and EQ-5D-5L at 3, 6, 9, and 12-months post-procedure. An interim analysis to assess for superiority was prespecified and overseen by an independent data management committee (DMC) when a minimum of 60% of patients had completed their 3-month primary endpoint visit.
The interim analysis showed clear statistical superiority (p < 0.001) for all primary and secondary patient-reported outcome measures in the RF ablation arm compared to ongoing standard care arm. This resulted in a DMC recommendation to halt enrollment in the study and offer early cross-over to the control arm. As a result, the study reported the outcomes of the 104 patients included in the intent-to-treat (ITT) analysis of the 3-month primary endpoint, which included 51 patients in the RF ablation arm and 53 patients in the standard care arm. At baseline, the mean age was 50 years, mean ODI was 46.1 (severe pain disability) and mean VAS was 6.67 cm (on a 0 to 10 cm scale). More than 67% of patients reported experiencing LBP for greater than 5 years and more than 70% had received prior injections at baseline.
Comparing the RF ablation arm to the standard care arm (Figure 2), the mean changes in ODI at three months were -25.3 points versus -4.4 points, respectively, resulting in an adjusted difference of 20.9 points (p<0.001); and mean changes in VAS were -3.46 versus -1.02, respectively, an adjusted difference of 2.44 cm (p<0.001) (Figure 3). In the RF ablation arm, 74.5% of patients achieved the minimal clinically important difference (MCID) of ≥ 10-point improvement in ODI, compared with 32.7% in the standard care arm (p < 0.001). With a MCID of 2.0 cm improvement in VAS, 72.5% of patients in the RF ablation arm reached clinical success compared to 34.0% of patients in the standard care arm. No RF ablation patients received a spinal injection prior to the 3-month endpoint, while in the standard care arm, 6 standard of care patients (11%) received injections across 5 study sites. The study concluded that minimally invasive RF ablation of the BVN leads to significant improvement of pain and function at 3-months in patients with suspected chronic vertebrogenic related LBP.
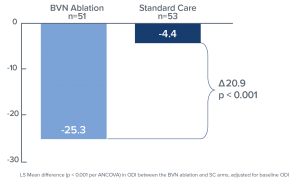
Figure 2. Bar graph demonstrating mean ODI changes at 3 months both for the BVN ablation and the standard care groups. Statistically significant improvement of the patients’ function is noted in the BVN group (p < 0.001).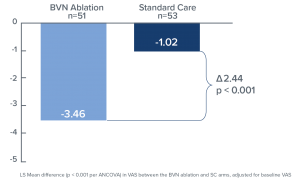
Figure 3. Bar graph demonstrating the mean difference in the VAS at 3 months both for the BVN Ablation and Standard Care groups. Statistically significant improvement in patients treated with BVN is noted (p < 0.001).
SMART Trial29:
The SMART trial was a prospective randomized, sham-controlled, double-blinded, FDA-IDE trial conducted to evaluate the safety and efficacy of RF ablation of the BVN for the treatment of CLBP. A total of 225 CLBP patients with Type I or Type II Modic changes noted in vertebral bodies L3 to S1 were randomized to either a sham-control (78 patients) or BVN ablation treatment (147 patients). All study participants were treated with the same operating protocol and pedicle access. The sham-control arm received simulated RF ablation therapy. Treatment success was adjudicated in a blinded review of the 6-week MRI. Study participants were followed at 2 weeks, 6 weeks, and 3, 6, 9, and 12 months post randomized intervention. The primary efficacy endpoint was change in ODI from baseline to 3 months post-procedure. The primary safety endpoint was a comparison of musculoskeletal and neurologic adverse events at 12 months.
Participants in this study were of working age (mean of 47 years), reported severe disability impact from their low back pain (mean ODI of 42), and more than 68% had been experiencing CLBP for greater than 5 years. At 3 months, the mean ODI in the treatment arm decreased 20.5 points, as compared to a 15.2 point decrease in the sham arm (p = 0.019, per-protocol population). The reduction in ODI experienced by the treatment arm was twice the minimally clinically important difference of ≥ 10 points and responder rates were 75.6% in the treatment arm compared to 55.3% in the sham control arm. There were no serious device or procedure-related adverse events reported in patients randomized to the RF ablation treatment arm through 12 months.
This level 1 trial demonstrated significant functional improvement in patients treated with RF ablation of the BVN for CLBP compared to patients treated with a sham procedure. Safety of the procedure was also demonstrated. The results supported BVN ablation as a minimally invasive treatment for the relief of chronic low back pain.
SMART 24 Month Outcomes30:
This prospective, single-arm study is an extension of follow-up for the RF ablation treatment arm of the SMART trial. Per the original SMART RCT protocol, at completion of the 12-month primary safety endpoint, patients in the sham-control arm could cross to BVN ablation treatment; 73% elected to cross. Due to this high rate of cross-over, the 147 RF ablation treatment arm participants acted as their own control in comparing 24-month outcomes to baseline.
Clinical improvements in the ODI, VAS, and the Medical Outcomes Trust Short-Form Health Survey Physical Component Summary (SF-36 PCS) were statistically significant compared to baseline at all follow-up time points through 2 years (3, 6, 9, 12, 18 and 24 months). The mean percent improvements at 2 years in ODI (Figure 4) and VAS compared to baseline were 53.7% and 52.9%, respectively. Responder rates for ODI and VAS were also maintained through 2 years for both a 10-point ODI MCID threshold (76.4% of patients) and an ODI 20-point improvement threshold (57.5% of patients); the MCID threshold for VAS of 1.5 cm improvement was reported in 70.2% of patients at 24 months (Figure 5). In summary, patients treated with RF ablation of the BVN for CLBP exhibited sustained clinical benefits in ODI and VAS and maintained high responder rates through 2 years following treatment.
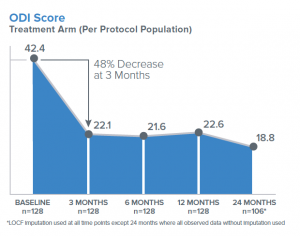
Figure 4. Mean ODI Score in per-protocol treatment arm followed up to 24 months.
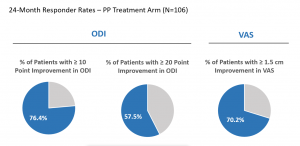
Figure 5. Shows the 24-Month ODI & VAS responder rate – per-protocol arm at 24 months (N=106).
Evidence and Literature Conclusion
Intraosseous ablation of the BVN is supported by a basic and clinical evidence foundation, including a Level I, sham controlled RCT and a second Level I RCT against standard conservative management. Data through 24 months suggests durability of the treatment effect. Collectively, these studies demonstrate that BVN ablation provides clinically meaningful improvements in pain and function to 2 years with an excellent safety profile. This evidence supports BVN as a treatment option for a well-defined sub-population of CLBP patients.
Indications for Surgery
Intraosseous ablation of the BVN from the L3 through S1 vertebrae may be considered medically indicated for individuals with chronic low back pain when all of the following criteria are met:
- Chronic axial low back pain (CLBP) of at least 6 months duration; and
- Failure to respond to at least 6 months of non-surgical management and
- MRI demonstrated Modic Type 1 or 2 changes in at least one vertebral endplate, at one or more levels from L3 to S1.
Coding and Coverage History
Intraosseous ablation of the basivertebral nerve is a new procedure not previously performed. As such, this procedure currently should be reported with CPT 22899 (unlisted procedure, spine).
CPT Code Description 22899 Unlisted procedure, spine Typical ICD-10 diagnosis codes that indicate medical necessity are:
ICD-10 Description M47.816 Spondylosis w/o myelopathy or radiculopathy, lumbar region M47.817 Spondylosis w/o myelopathy or radiculopathy, lumbosacral region M51.36 Other intervertebral disc degeneration, lumbar M51.37 Other invertebral disc degeneration, lumbo-sacral M54.6 Low back pain HCPCS (effective date: Jan. 1, 2019) Description C9752 Destruction of intraosseous basivertebral nerve, first two vertebral bodies, including imaging guidance (e.g., fluoroscopy), lumbar/sacrum C9753 Destruction of intraosseous basivertebral nerve, each additional vertebral body, including imaging guidance (e.g., fluoroscopy), lumbar/sacrum (list separately in addition to code for primary procedure) Conclusion
Intraosseous ablation of the basivertebral nerve is a relatively new minimally invasive treatment for the relief of CLBP that is diagnosed using well-established clinical and MR imaging findings. The procedure is supported by Level I evidence including 2 RCTs demonstrating a statistically significant decrease in pain and an improvement in function with outcomes sustained to at least 24 months in a limited number of studies. These results were seen in a patient population that is one of the most expensive and difficult to provide care for and in this era of rising healthcare costs and increasing need for therapies to reduce the use of opioids, BVN ablation may provide a treatment option to fill the gap in the treatment paradigm for patients that fail non-surgical treatment.
Limitations
- Industry funding is a potential source of study bias for the available data reviewed.
- Limited number of studies.
- Short-term follow-up for the majority of studied patients.
- Unknown effect on the primary degenerative process.
References
- Deyo RA, Mirza SK, Martin BI. Back pain prevalence and visit rates: estimates from U.S. national surveys, 2002. Spine (Phila Pa 1976) 2006;31:2724-7.
- Frymoyer JW. Back pain and sciatica. N Engl J Med 1988;318:291-300.
- Hoy D, Bain C, Williams G, et al. A systematic review of the global prevalence of low back pain. Arthritis Rheum 2012;64:2028-37.
- Thiese MS, Hegmann KT, Wood EM, et al. Prevalence of low back pain by anatomic location and intensity in an occupational population. BMC Musculoskelet Disord 2014;15:283.
- CDC Summary of Health Statistics: National Health Interview Survey.
- Knauer SR, Freburger JK, Carey TS. Chronic low back pain among older adults: a population-based perspective. J Aging Health 2010;22:1213-34.
- Meucci RD, Fassa AG, Faria NM. Prevalence of chronic low back pain: systematic review. Rev Saude Publica 2015;49.
- Shmagel A, Foley R, Ibrahim H. Epidemiology of Chronic Low Back Pain in US Adults: Data From the 2009-2010 National Health and Nutrition Examination Survey. Arthritis Care Res (Hoboken) 2016;68:1688-94.
- Freburger JK, Holmes GM, Agans RP, et al. The Rising Prevalence of Chronic Low Back Pain(). Arch Intern Med 2009;169:251-8.
- Johannes CB, Le TK, Zhou X, et al. The prevalence of chronic pain in United States adults: results of an Internet-based survey. J Pain 2010;11:1230-9.
- Dagenais S, Caro J, Haldeman S. A systematic review of low back pain cost of illness studies in the United States and internationally. Spine J 2008;8:8-20.
- Luoma K, Riihimaki H, Luukkonen R, et al. Low back pain in relation to lumbar disc degeneration. Spine (Phila Pa 1976) 2000;25:487-92.
- Cheung KM, Karppinen J, Chan D, et al. Prevalence and pattern of lumbar magnetic resonance imaging changes in a population study of one thousand forty-three individuals. Spine (Phila Pa 1976) 2009;34:934-40.
- Livshits G, Popham M, Malkin I, et al. Lumbar disc degeneration and genetic factors are the main risk factors for low back pain in women: the UK Twin Spine Study. Ann Rheum Dis 2011;70:1740-5.
- Brown MF, Hukkanen MV, McCarthy ID, et al. Sensory and sympathetic innervation of the vertebral endplate in patients with degenerative disc disease. J Bone Joint Surg Br 1997;79:147-53.
- Antonacci MD, Mody DR, Heggeness MH. Innervation of the human vertebral body: a histologic study. J Spinal Disord 1998;11:526-31.
- Modic MT, Steinberg PM, Ross JS, et al. Degenerative disk disease: assessment of changes in vertebral body marrow with MR imaging. Radiology 1988;166(1 Pt 1):193-9. DOI: 10.1148/radiology.166.1.3336678
- Dudli S, Fields AJ, Samartzis D, et al. Pathobiology of Modic changes. Eur Spine J 2016;25:3723-34.
- Lotz JC, Fields AJ, Liebenberg EC. The Role of the Vertebral End Plate in Low Back Pain. Global Spine J 2013;03:153-64.
- Toyone T, Takahashi K, Kitahara H, Yamagata M, Murakami M, Moriya H. Vertebral bone-marrow changes in degenerative lumbar disc disease. An MRI study of 74 patients with low back pain. J Bone Joint Surg Br. 1994 Sep;76(5):757-64.
- Ulrich JA, Liebenberg EC, Thuillier DU, et al. ISSLS prize winner: repeated disc injury causes persistent inflammation. Spine (Phila Pa 1976) 2007;32:2812-9.
- Dudli S, Sing DC, Hu SS, et al. ISSLS PRIZE IN BASIC SCIENCE 2017: Intervertebral disc/bone marrow cross-talk with Modic changes. Eur Spine J 2017;26:1362-73.
- Georgy M, Stern M, Murphy K. What Is the Role of the Bacterium Propionibacterium acnes in Type 1 Modic Changes? A Review of the Literature. Can Assoc Radiol J. 2017 Nov;68(4):419-424. doi: 10.1016/j.carj.2017.07.004. Epub 2017 Oct 3.
- Kjaer P, Korsholm L, Bendix T, et al. Modic changes and their associations with clinical findings. Eur Spine J 2006;15:1312-9.
- Hoopes, PJ, et al Radiofrequency abloation of the basivertebral nerve as potential treatment of back pain: pathologic assessment in a bovine model. Proceedings of SPIE, 2005. 5698: p.168-180.
- Becker S, Hadjipavlou A, Heggeness MH. Ablation of the basivertebral nerve for treatment of back pain: a clinical study. Spine J 2017;17:218-23.
- Truumees E, Macadaeg K, Pena E, et al. A Prospective, Open –Label, Single-Arm, Multi-Center Study of Intraosseous Basivertebral Nerve Ablation For The Treatment of Chronic Low Back Pain. European Spine Journal 2019, https://doi.org/10.1007/s00586-019-05995-2
- Khalil J, Smuck M, Koreckij T, Keel J, Beall D, Goodman B, Kalapos P, Nguyen D, Garfin S. A Prospective, Randomized, Multi-Center Study of Intraosseous Basivertebral Nerve Ablation for the Treatment of Chronic Low Back Pain. Spine J. 2019 Jun 20. pii: S1529-9430(19)30800-9. doi: 10.1016/j.spinee.2019.05.598
- Fischgrund JS, Rhyne A, Franke J, et al. Intraosseous basivertebral nerve ablation for the treatment of chronic low back pain: a prospective randomized double-blind sham-controlled multi-center study. Eur Spine J. 2018;27(5):1146-1156. DOI: 10.1007/s00586-018-5496-1
- Fischgrund JS, Rhyne A, Franke J, Sasso R, Kitchel S, Bae H, et al. Intraosseous basivertebral nerve ablation for the treatment of chronic low back pain: two-year results from a prospective randomized double-blind sham-controlled multi-center Study. Int J Spine Surg 2019;13(2).
Disclosures and COI: Morgan Lorio, MD, has nothing to disclose; Olivier Clerk, MD, has nothing to disclose; Douglas Beall, MD, DAAPM, FIPP, reports the following disclosures: Paid consultant to: Medtronic, Spineology, Merit Medical, Lilly, Johnson & Johnson, SpinTech, Imaging3, IZI, Medlantis, Techlamed, Consultant, Peterson Enterprises, Medical Metrics, Radius Pharmaceuticals, Avanos, Vertiflex, Sollis Pharmaceuticals, Simplify Medical, Stryker, Lenoss Medical, Spine BioPharma, Piramal, ReGelTec, Nanofuse. Research funding from: Medtronic, SpinTech, Medical Metrics, Avanos, Relievant, Vertiflex, Stryker, Sollis Pharmaceuticals, Simplify Medical, Lenoss Medical, Spine BioPharma. Advisory Board member: Medtronic, Imaging3, ReGelTec, Nanofuse; Board member of: SpinTech, Nocimed. Stock or stock options: Artio, Sophiris, Eleven Biotherapeutics, Radius Pharmaceuticals, Flow Forward, Lenoss Medical, Spine BioPharma. Editorial support: Thieme, Springer, Humana. Speakers Bureau: Eli Lilly, Radius Pharmaceuticals, Stryker, Medtronic, Vivex, Vertiflex, Merit, Medlantis, Avanos, Piramal. Terrence Julien, MD, reports the following disclosures: Consulting: Medtronic, Stryker Spine, Stryker Navigation, Camber Spine/IMSE, Aurora Spine, Ulrich, RTI Surgical, Spinal Elements, BioVentus Surgical, CoreLink, Novabone. Stock or stock options: Aurora Spine. Advisory Board member: Ulrich, RTI Surgical.
